Show summary Hide summary
- MIT gut protein discovery rewrites mucus defense strategy
- Dual-function immune response against resistant gut pathogens
- Intelectin‑2 and intestinal health: striking the right balance
- Next-gen antimicrobials inspired by your own gut protein
- Key takeaways you can remember and share
- What exactly is intelectin‑2 and where is it found?
- How does this gut protein capture and eliminate harmful bacteria?
- Can intelectin‑2 affect beneficial microbes in the microbiome?
- Why is this discovery important for antibiotic resistance?
- Are there therapies using intelectin‑2 available today?
You already host a microscopic bodyguard that can capture and eliminate harmful bacteria before they invade your tissues. MIT researchers have just revealed how this hidden intelectin 2 gut protein pulls off a double defensive move inside the intestine.
By decoding how this lectin behaves in the gastrointestinal tract, the team shows that your mucus barrier is far more active than a simple “slime coat.” This discovery links everyday intestinal health, the microbiome, and the rising challenge of antibiotic resistance in a surprisingly concrete way. To learn more about how inflammation is controlled, visit scientists uncover body’s key to controlling inflammation.
MIT gut protein discovery rewrites mucus defense strategy
The star of the study, intelectin‑2, sits on the moist surfaces of the gut, exactly where microbes first make contact with your body. Unlike many immune molecules that only signal danger, this protein acts directly. It recognizes specific sugars on bacterial surfaces and on mucus molecules, then uses them as docking points.
Scientists Uncover the Unexpected Truth About How Human Hair Actually Grows, Challenging Textbook Beliefs
How Serotonin-Producing Gut Bacteria Could Unlock New Treatments for IBS
MIT researchers found that intelectin‑2 behaves like a biochemical Velcro strip. It latches onto sugar motifs, pulls bacteria into the mucus network, slows their growth, and helps keep them away from the intestinal lining. This places the protein at the crossroad between physical barrier, immune response, and microbial control.
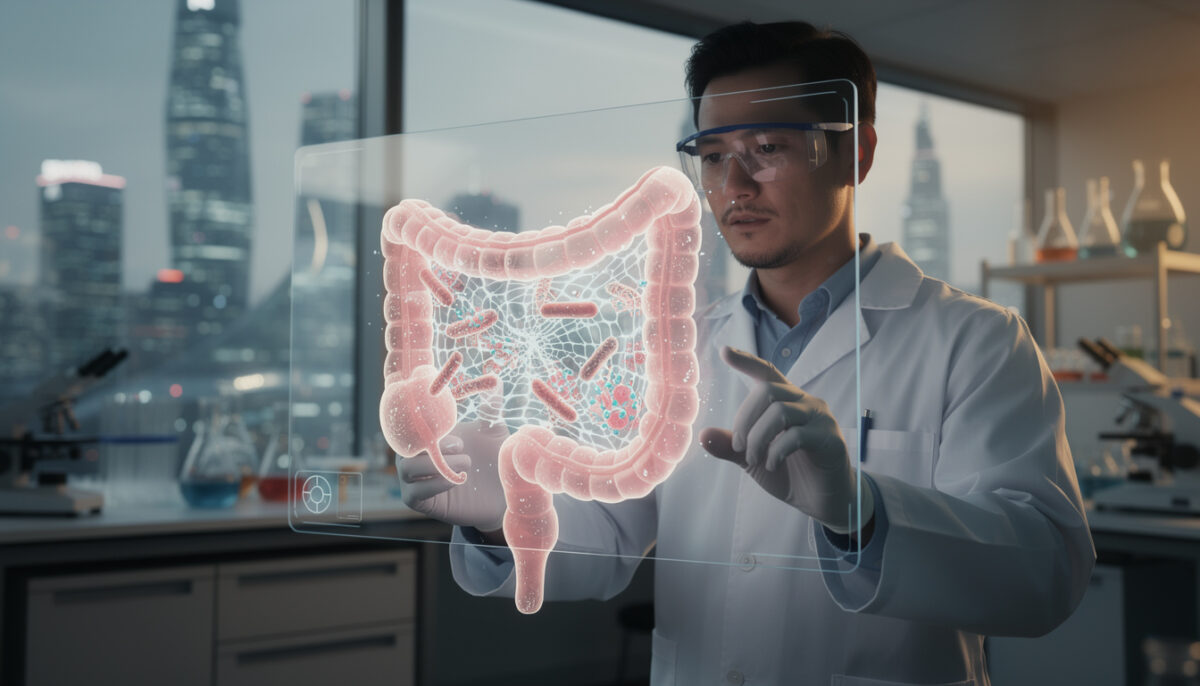
How intelectin‑2 locks bacteria into the mucus layer
Intelectin‑2 belongs to the family of lectins, proteins specialized in reading sugar “barcodes” on cells and microbes. The team showed that intelectin‑2 binds strongly to galactose, a sugar frequently present in mucins, the long molecules that form the mucus gel. By connecting multiple mucins together, it tightens the mesh and thickens the barrier that protects your intestinal wall.
The same galactose motifs also decorate the surface of several bacterial species. Once intelectin‑2 attaches to these microbes, they become physically trapped in the mucus and gradually start to fall apart, indicating that their membranes are destabilized. That dual action turns a passive coating into an active antimicrobial scaffold. Explore how scientists unveil bacterial kill switch are affecting the fight against superbugs.
Dual-function immune response against resistant gut pathogens
The work, detailed in outlets such as MIT News on GI tract proteins, highlights how intelectin‑2 targets a broad panel of microbes. Tests showed activity against pathogens commonly involved in gut infections, including strains of Staphylococcus aureus and Klebsiella pneumoniae that often withstand standard antibiotics. The protein does not rely on a single molecular trick, which complicates bacterial escape.
First, intelectin‑2 reinforces the mucus shield, making it harder for invaders to approach the epithelial surface. Then, if some bacteria start to break through, the protein binds their surface sugars, restrains them in place, and eventually disrupts them. This layered defense gives your body extra time before inflammation or infection escalates.
Human and mouse intestines, same protein, different tactics
The team also uncovered how different organisms deploy the same molecule. In humans, Paneth cells of the small intestine constantly produce intelectin 2 gut protein, creating a steady antimicrobial presence. In mice, the protein comes mainly from mucus‑secreting Goblet cells and is switched on in response to inflammation or certain parasites.
This contrast shows that evolution tunes the same lectin to local needs. Continuous production favors a stable barrier in humans, while inducible production in mice creates a rapid-response tool. For researchers developing biotechnology applications, that flexibility opens several design options for future therapies. Get more insights from a monthly aging fish unveils the secrets of kidney aging.
Intelectin‑2 and intestinal health: striking the right balance
The findings matter directly for conditions where the mucus barrier is weakened, especially inflammatory bowel disease. In these patients, intelectin‑2 levels can be unusually low or, on the contrary, excessively high. Too little and the barrier loosens, making the epithelium more accessible to pathogens. Too much and the protein can wipe out helpful microbes that contribute to a resilient microbiome.
Restoring the correct range becomes a therapeutic target. The study aligns with broader work on barrier repair, such as research on proteins that may help mend “leaky gut” and modulate mood, highlighted in articles like emerging leaky-gut protein research. Intelectin‑2 now enters this conversation as a powerful regulator of mucus structure and microbial composition.
What this means for everyday microbiome care
The story of intelectin‑2 reminds you that gut defense is not only about killing germs; it is about structure, timing, and cooperation with resident microbes. A stable mucus layer lets beneficial bacteria thrive in the outer regions, while lectins like intelectin‑2 keep invasive strains at bay and maintain order along the epithelium.
For a reader like Daniel, an amateur cyclist trying to optimize recovery and digestion, this translates into a new lens on diet and lifestyle. Supporting mucus integrity and lectin activity through balanced nutrition, fiber intake, and medical follow-up when symptoms appear could protect performance and comfort on and off the bike.
Next-gen antimicrobials inspired by your own gut protein
Because intelectin 2 gut protein targets sugars rather than classical antibiotic pathways, it offers a fresh template against resistant microbes. The protein recognizes motifs that many bacteria cannot easily discard without losing vital functions. That makes lectin-inspired molecules attractive candidates for the next generation of antimicrobial tools rooted in human biology.
Some groups already explore how lectin engineering can reinforce mucus barriers or selectively block pathogens, as discussed in reports like chemistry-focused analyses of gut lectins and broader summaries on platforms such as ScitechDaily’s coverage of natural bacterial traps. Intelectin‑2 becomes a blueprint rather than just a curiosity.
Key takeaways you can remember and share
If you had to explain this discovery over coffee to a friend who loves science podcasts, four points would stand out. They connect everyday digestion with cutting-edge immunology in a way that is easy to visualize and hard to forget.
- Intelectin‑2 is a dual‑function gut protein: it both strengthens the mucus barrier and directly neutralizes or restrains harmful bacteria.
- It reads sugar codes on mucus and microbes, using galactose motifs as anchors to capture and immobilize unwanted bacteria.
- Mucus is an active immune surface, not passive slime; its architecture and lectins shape your intestinal health and microbiome composition.
- Lectin‑based strategies could inspire new therapies against antibiotic‑resistant infections while helping repair damaged barriers in chronic gut diseases.
These insights turn a microscopic protein into a tangible ally, reshaping how you can think about protection along every centimeter of your gastrointestinal tract.
What exactly is intelectin‑2 and where is it found?
Intelectin‑2 is a carbohydrate‑binding lectin produced in the gastrointestinal tract. In humans, Paneth cells of the small intestine secrete it into the mucus layer. There it binds sugars on mucins and on microbes, helping both to stabilize the mucus barrier and to restrain potentially harmful bacteria before they reach the intestinal lining.
How does this gut protein capture and eliminate harmful bacteria?
Intelectin‑2 recognizes specific sugar structures, including galactose, displayed on bacterial membranes. By binding these sugars, it traps microbes within the mucus mesh and slows their growth. Over time, the interaction disrupts bacterial membranes, which leads to their breakdown and reduces the risk of infection or inflammation in the surrounding tissue.
Can intelectin‑2 affect beneficial microbes in the microbiome?
Yes, extremely high levels of intelectin‑2 may also hit beneficial bacteria that normally support digestion and immune balance. The study suggests that intestinal health depends on keeping intelectin‑2 within a physiological range, strong enough to stop pathogens but not so aggressive that it erodes the diversity and stability of the resident microbiome.
Why is this discovery important for antibiotic resistance?
A Serendipitous Lab Error at Cambridge Uncovers a Groundbreaking Technique to Transform Drug Molecules
AI-Powered Toys Are Here to Stay, But Their Safety Remains Uncertain
Intelectin‑2 attacks bacteria through sugar recognition instead of the classic antibiotic targets inside cells. Some strains of Staphylococcus aureus and Klebsiella pneumoniae, which show resistance to multiple drugs, remain vulnerable to this lectin‑based mechanism. Designing treatments that mimic or boost intelectin‑2 could offer new antimicrobial options when traditional antibiotics fail.
Are there therapies using intelectin‑2 available today?
Current work remains at the research stage, with no approved drugs directly based on intelectin‑2 yet. However, the findings already guide projects in biotechnology that aim to reinforce the mucus barrier, tune lectin levels in inflammatory bowel disease, and develop lectin‑inspired molecules as future antimicrobial treatments aligned with the body’s own immune response.