Show summary Hide summary
- Why Frozen Organs Have Always Been the Impossible Dream
- The Science That Finally Cracked the Problem
- Are We Ready for a Future with Unlimited Organs?
- What Could Still Go Wrong? The Doubts No One’s Talking About
- Could This Shift the Balance in Who Gets to Live—And How?
- FAQ
- How does this new organ cryopreservation method prevent cracking during freezing?
- Could organ cryopreservation extend how long organs can be stored for transplants?
- What challenges remain before cryopreserved organs can be used in hospitals?
- Will organ cryopreservation impact organ transplant waiting lists?
- Are there risks or side effects to receiving a cryopreserved organ?
Picture the moment: a team of scientists has just discovered how to freeze transplant organs without cracking them, a challenge that has haunted medicine for decades. Not in theory or in distant animal trials, but with real tissues, held at subzero temperatures with no shattered cells and no fatal damage. What does it mean when human hearts, kidneys, and livers might soon be placed on ice, indefinitely paused, and then revived as fresh as the day they were donated? how to freeze transplant organs without cracking them
This breakthrough could shatter the chilling barrier that has kept organ shortages at crisis levels and changed the rules about who gets a second chance at life. Yet as this promise of unlimited organs moves from science fiction into surgical suites, it also ushers in a new era of uncomfortable questions. Who will get to benefit, and what unforeseen consequences could ripple through medicine, ethics, and society? The ice may finally have been tamed, but the story is just beginning.
Why Frozen Organs Have Always Been the Impossible Dream
For decades, the idea of cryopreservation for organ transplantation sounded almost too good to challenge. In theory, freezing human organs could have erased the brutal realities of the organ shortage: no more hours spent racing donated kidneys or hearts across continents, no more heartbreak from missed matches. Yet every attempt collided with the same, stubborn wall—ice crystal damage. When organs are frozen using traditional methods, jagged crystals form and tear apart delicate cellular structures. Instead of keeping tissue alive, freezing has reliably destroyed it.
Did the human eye really evolve from an ancient ‘cyclops’? The strange story science textbooks missed
Scientists created a plastic that kills viruses instantly—here’s what actually happens when you touch it (and what they’re not sure about yet)
- Researchers have tried countless workarounds, from antifreeze-like chemicals to ultra-rapid cooling, but the result is almost always the same: tissue ruined before it can ever save a life.
- The science was clear and unsparing—until now.
- The limitations of cryopreservation have entrenched a system where viable organs are measured in hours, not days. The limitations of cryopreservation
- Surplus organs perish on ice, and patients can wait years for a chance at survival.
- With so much riding on a breakthrough, any real solution is bound to raise urgent new questions.
The Science That Finally Cracked the Problem
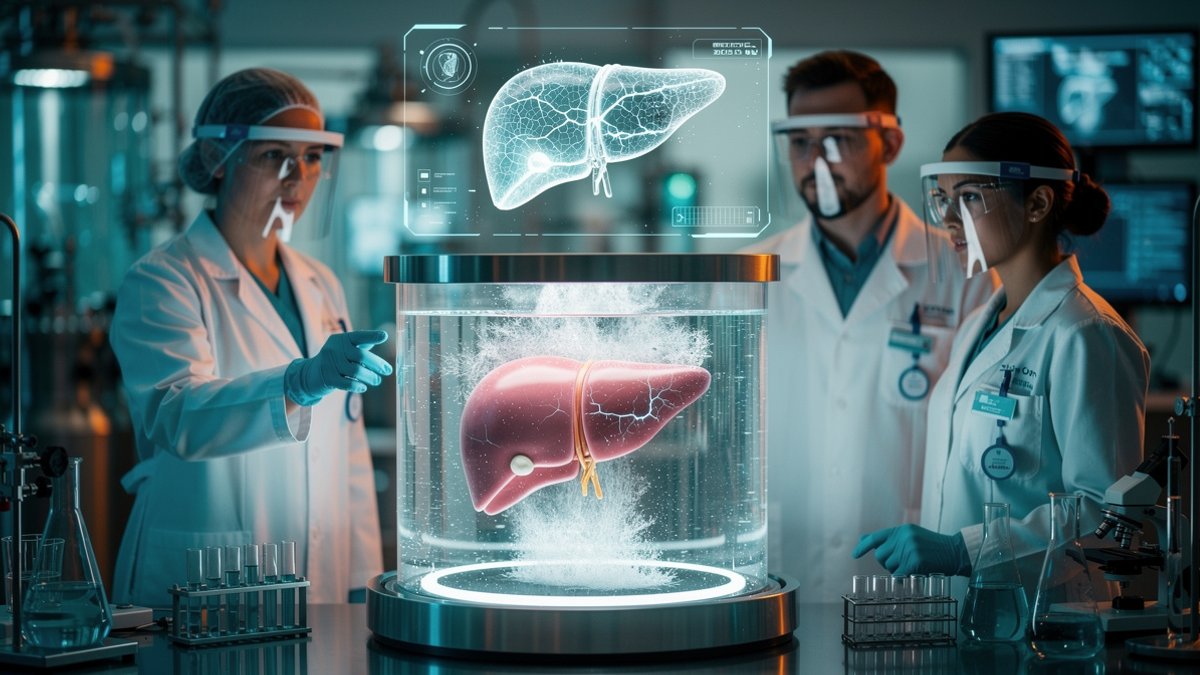
For decades, freezing an organ meant an inevitable death sentence for its cells. Ice crystals would burst delicate membranes, and lethal cracks would spider through tissue during the thaw. But a new blend of cryoprotectant chemicals and meticulously timed cooling has upended these rules. The breakthrough lies in shifting from traditional slow freezing to a process called vitrification, which transforms living tissue into a glass-like state. In this state, water molecules never form damaging ice but are locked in place, vastly reducing thermal stress and internal ruptures.
- Achieving true vitrification was long dismissed as impractical.
- Too little cryoprotectant and you still get lethal ice; too much, and the organ itself is poisoned.
- The secret, researchers found, is a rapid yet controlled freezing protocol.
- By cooling tissues at precisely calculated rates and concentrations, organs now solidify uniformly, sidestepping the catastrophic fractures of the past.
- Unexpectedly, not just the chemistry but the choreography of temperature and timing seems paramount. rapid yet controlled freezing protocol
This isn’t just a technical tweak. The result is organs that survive deep-freeze intact, opening doors to long-term storage and global transport. Yet, as promising as this sounds, the shift from theory to human application comes loaded with new unknowns and risks few are yet addressing. nasal spray shows promise
Are We Ready for a Future with Unlimited Organs?
If scientists can stock organ banks with flawless frozen organs, the logistics of transplant medicine will shift radically. Suddenly, the old race against the clock might be replaced by a new challenge: who decides how to distribute this unprecedented bounty? For patients stuck on waitlists, this could mean hope—unless the allocation system fails to keep pace with the technology.
With abundance, cracks appear elsewhere. Could hospitals in wealthier regions corner supplies, pushing transplant equality further out of reach? Or will the bioethics of this breakthrough spur fierce debate about who deserves priority—and who gets left behind? Logistics loom large, too. Storing, tracking, and transporting thousands of viable organs will force hospitals to reinvent their infrastructure just to keep up.
- The gleam of unlimited organs is real, but so is the risk of new inequities and unforeseen divides.
- The science is dazzling.
- The human system around it may not be ready. top must read
What Could Still Go Wrong? The Doubts No One’s Talking About
Yet, for all its promise, this breakthrough leaves scientists staring down a host of uncomfortable uncertainties. Thawing an organ without cracks is only the beginning. Can its cells truly recover full function, or do hidden injuries lurk beneath the surface? Long-term tissue viability remains largely untested—no one knows how well these organs will actually perform after months or years on ice.
There is also the risk of unforeseen complications. Subtle changes during freezing and thawing could spark immune responses or long-term organ dysfunction, problems that simply do not show up in short-term lab studies. Until rigorous medical trials and exhaustive functional testing prove otherwise, clinical uncertainty will cast a long shadow. Surgeons and patients alike will want more than scientific optimism—they will want proof that a frozen organ means a living future, not just a technical milestone.
Could This Shift the Balance in Who Gets to Live—And How?
If freezing organs becomes standard, the game around transplant prioritization changes overnight. The old constraints—matching a donor’s last heartbeat to a recipient within hours—could vanish. Suddenly, allocation policy might matter more than luck or geography. But with more organs in the queue, who decides who moves to the front? What about those already left behind by biases in the system?
Medical ethics will be stress-tested. Advances in future medicine tend to arrive before consensus catches up, and history is littered with cautionary tales. Who will set the rules for this new playing field—the same committees, or someone else entirely? If the technology delivers as promised, it could flatten disparities but also shift power in unsettling ways.
This breakthrough dares us to ask not just how many lives can be saved, but whose lives society values most when scarcity no longer sets the pace. The organ shortage might become a relic, but ethical uncertainty is just getting thawed out.
FAQ
How does this new organ cryopreservation method prevent cracking during freezing?
The breakthrough technique uses specialised solutions and precise cooling rates to avoid the formation of ice crystals, which normally cause cracks and damage in organs. This allows tissues to be frozen without the usual cell destruction.
Could organ cryopreservation extend how long organs can be stored for transplants?
Yes, successful organ cryopreservation could allow organs to be stored for much longer periods, potentially transforming transplant logistics and dramatically reducing waste from expired donations.
What challenges remain before cryopreserved organs can be used in hospitals?
Researchers need to confirm that the freezing and revival process works reliably for all organ types and does not impact long-term function. Regulatory approval, ethical considerations, and scaling the technology for routine use are still ahead.
Will organ cryopreservation impact organ transplant waiting lists?
Golden oyster mushrooms spark urgent scientist warnings in Florida—what could be lurking in your local market pick?
Scientists Find Bacteria Can ‘Explode’—And This Shocking Twist Could Be Supercharging Antibiotic Resistance
If widely adopted, organ cryopreservation could reduce waiting times by making more organs available and matching them more effectively with recipients, potentially saving many more lives.
Are there risks or side effects to receiving a cryopreserved organ?
While the new method aims to avoid cell damage, scientists are still studying whether subtle effects could appear after transplantation or during long-term follow-up, making careful monitoring essential as trials progress.


